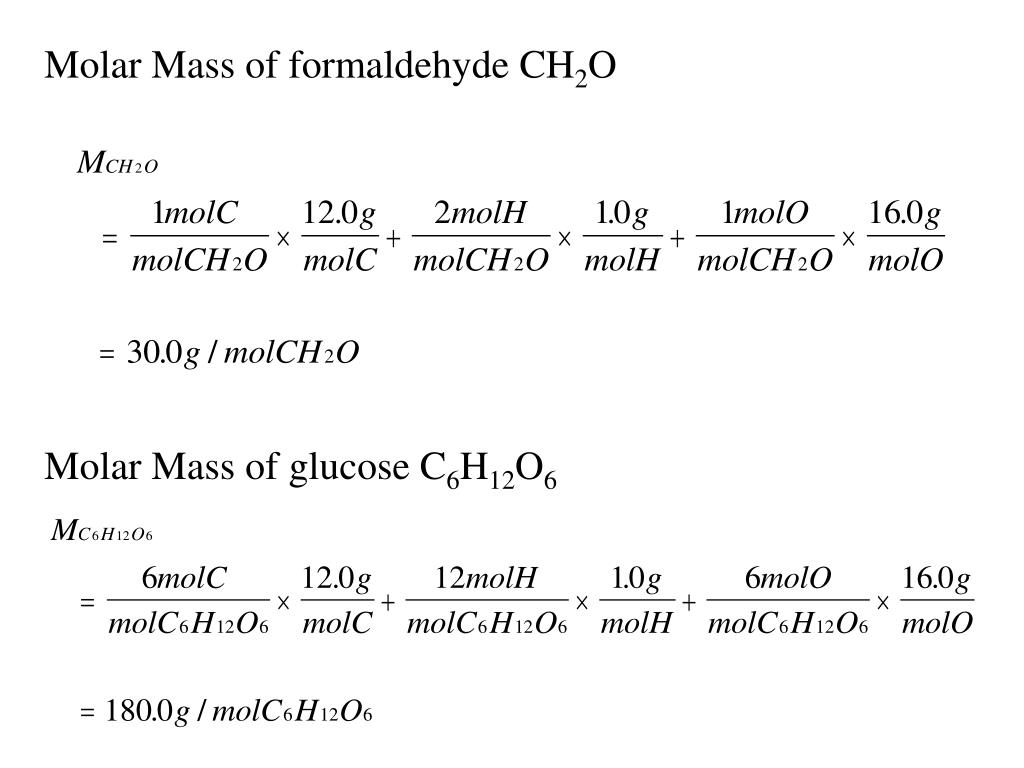
Molar Mass Ch2O. The mass of ch2o is 12 + 2*1 + 16 = 30. The empirical formula for a compound is ch2o and the molar mass is 180.2g mol which is molecular formula for t… get the answers you need, now!
A compound with the empirical formula ch2o has a molar mass of approximately 90 g/mol. 👍 👎 👁 jane jun 11, 2020 empirical formula is ch2o. This means that you can think about the empirical formula as being a sort of building block for the molecule.
The Given Chemical Compound Has 2 Atoms Of Hydrogen And One Atom Of.
👍 👎 👁 jane jun 11, 2020 empirical formula is ch2o. • co2 + 4h2s + o2 = ch2o + 4s +. See also our theoretical yield calculator for chemical reactions (probably your next stop to finish the problem set).
Number Equal To The Sum Of The Atomic Masses Of The Atoms In A Molecule.
A compound with the empirical formula ch2o has a molar mass of approximately 90 g/mol. The molecular weight of the compound is 180.18 which is approximately 180. Guest feb 10, 2017 2 answers #1 +35128 0 the molar mass is given as 30.03 gm / mol you have 4.55 moles just multiply them together 30.03 gm/mol x 4.55 mol = 136.64 gms in 4.55 moles ch2o electricpavlov feb 10, 2017 #2 +35128 0
Molar Mass Of (Ch2O)6 Is 180.1559 G/Mol
This means that you can think about the empirical formula as being a sort of building block for the molecule. Molar mass of ch2o molar mass of ch2o is 30.0260 g/mol compound name is formaldehyde forget 2020. Convert grams ch2o to moles or moles ch2o to grams.
3*12 = 36 6*1 = 6 3*16 = 48 48 + 6 + 36 = 90 👍 👎 👤 Drbob222 Feb 27, 2020 Respond To This Question Similar Questions Chemistry
Check the chart for more details. A compound with the empirical formula ch2o has a molar mass of approximately 90 g/mol. The molar mass of carbon is 12, the molar mass of hydrogen is 1 and the molar mass of oxygen is 16.
Molar Mass Of Ch2O = 30.02598 G/Mol.
The mass of ch2o is 12 + 2*1 + 16 = 30. The empirical formula of a chemical compound is ch2o. Empirical mass x #units = molar mass 30*units = 90 so you must have (ch2o)3 or c3h6o3.