Draw The Lewis Structure For Hcch Ethyne. There are no lone pairs on carbon or hydrogen atoms. Hcch lewis structure polar or nonpolar.
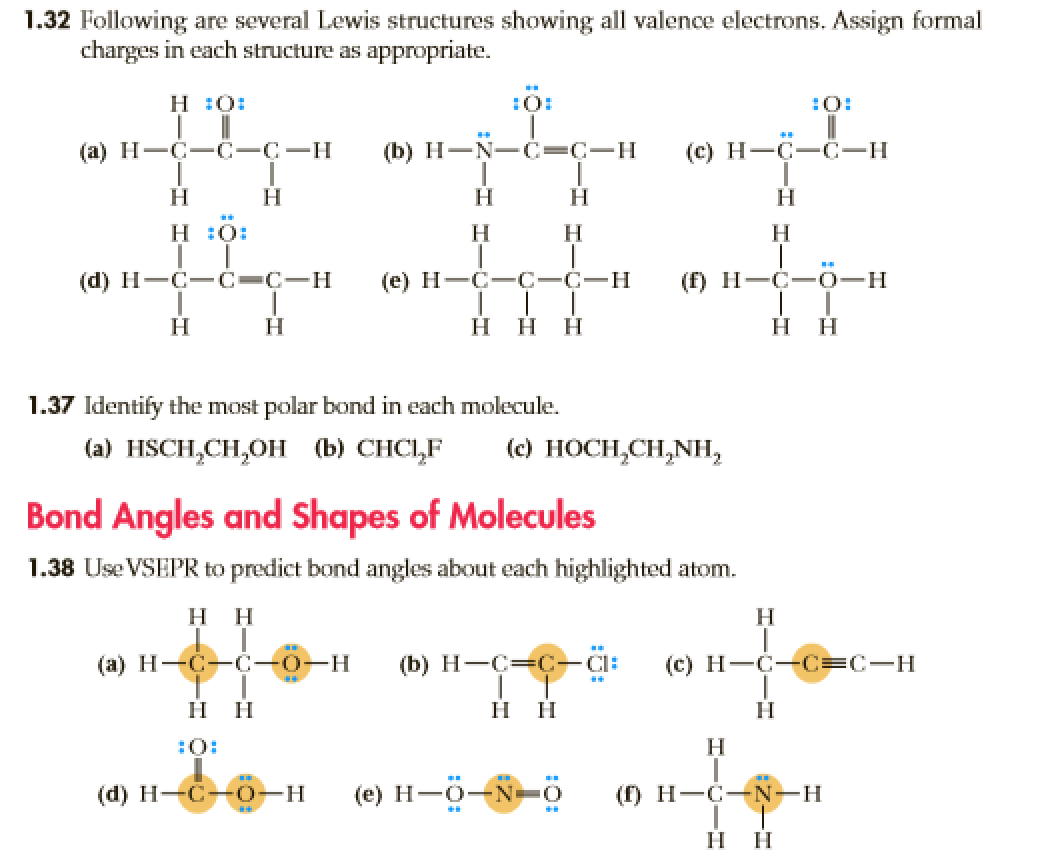
Draw the lewis structure for hcch hcch (ethyne). Draw the lewis structure for hcch (ethyne). C2h2 lewis structure tutorial how to draw the for ethyne or acetylene note, hydrogen atoms (h) should not have lone pair.
Ethyne Has A Triple Bond Between The Two Carbon Atoms.
One additional one could get some one minus charge. Include all hydrogen atoms and all lone pairs of electrons reset ? Mar 26 2021 05:54 am.
Acetylene Was Found In Whole Gas Octane Levels 87, 89 And 92 At 0.0022, 0.0032 And 0.0037 Ppbc%, Respectively (1).
Hcch (ethyne) can also be written as c 2 h 2. Lewis structure for c2h2 (ethyne) commonly tested lewis structures we draw lewis structures to predict: 70 more lewis dot structures.
The First Thing We Need To Do Is Some Of All The Violence.
For the hcch lewis structure you'll need to form a triple bond between the two carbon atoms. Draw the molecule by placing atoms on the grid and connecting them with bonds. This molecule is also known by the name acetylene.
Part A Draw The Lewis Structure For Hcch (Ethyne).
The compound has a simple structure and is made up of two carbon atoms and two hydrogen atoms. So that gives us eight valence electrons. Then the octet is satisfied for oxygen and the duet for hydrogen.
Acetylene Has Been Detected 12%, 2.9% And 50% Of Total Hydrocarbon Concentration In Emissions From Vehicle Exhaust, Petroleum Exhaust And Petrochemical Plants, Respectively (2).
Acetylene is an unsaturated hydrocarbon with a triple bond. C 2 h 2 (acetylene | ethyne) lewis structure c 2 h 2 (acetylene or ethyne) contains two carbon atoms and two hydrogen atoms. They follow the duet rule (2 electrons).