Br Full Electron Configuration. The electron configuration is a system of notation that describes the distribution of all electrons in an atom. Home › what is br full electron configuration.
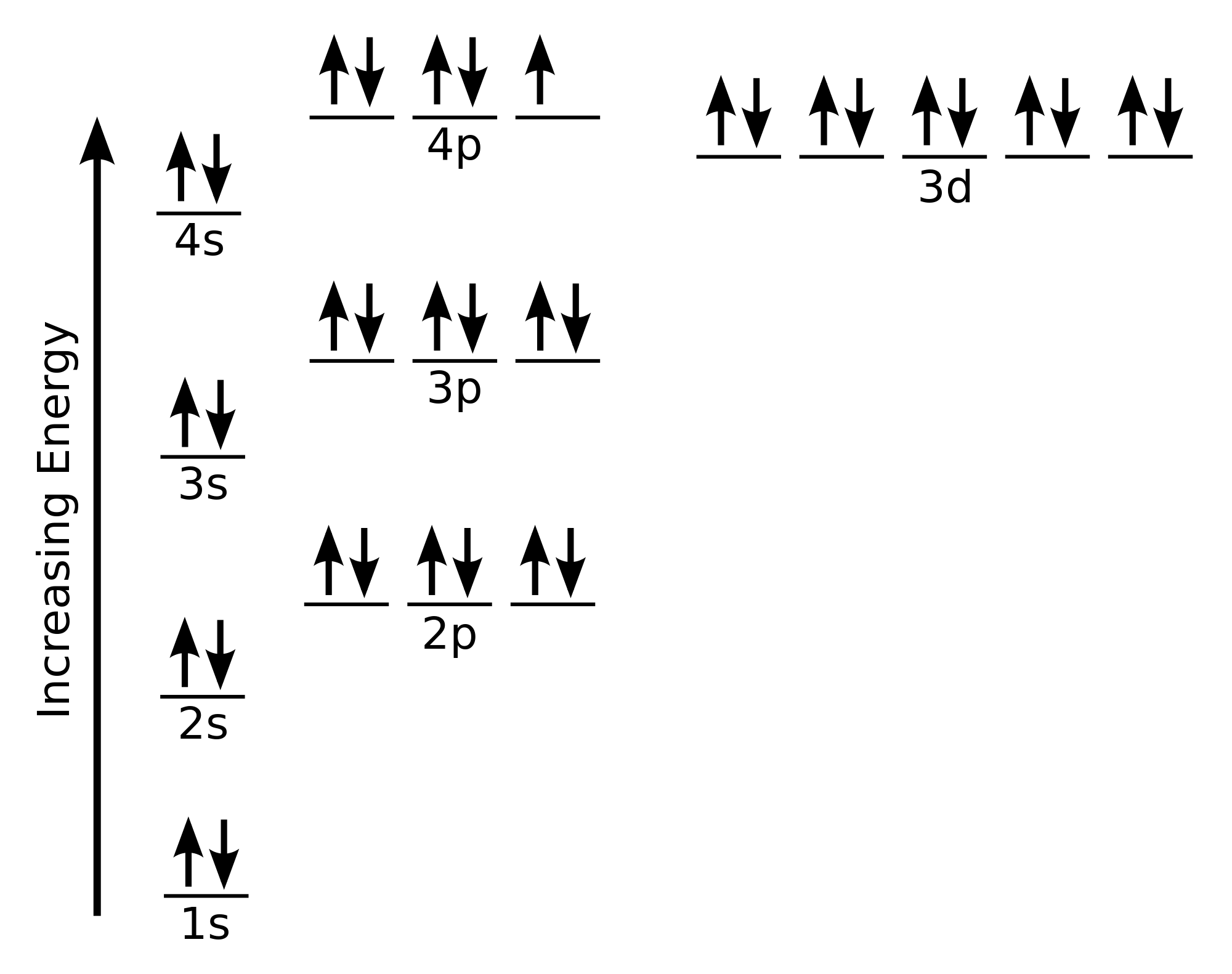
Bromine has the atomic number 35. 1s2 2s2 2p6 3s2 3p6 3d10 4s2 4p5. The element bromine was discovered by antoine jérôme balard in year 1826 in france.
Bromine Is A Chemical Element With Atomic Number 35 Which Means There Are 35 Protons And 35 Electrons In The Atomic Structure.
Many general rules are taken into consideration when assigning the location of the electron to its prospective energy state, however these assignments are arbitrary and it is always uncertain as to which electron is being The maximum number of electrons that can be accommodated by a subshell is given by the formula 2* (2l + 1). Write out the full electron configuration for each atom.
Niobium Overview Niobium Complete Electron Configuration 1S2 2S2 2P6 3S2 3P6 4 S2 3 D10 4 P6 5 S1 4 D4 Abbreviated Electron Configuration [Kr] 4D4.
To save room, the configurations are in noble gas shorthand.this means part of the electron configuration has been replaced with the element symbol of the noble gas symbol. Carbon has four valence electrons and here a value of four. In the case of b r x − it is an anion of charge minus 1 meaning that it will have one more electron than a normal bromine atom.
Since The Number Of Protons And Electrons Are Equal, We Can Identify The Electron Configuration By Filling The Orbitals With 35 Electrons.
Rb+ electron configuration (rubidium ion)in this video we will write the electron configuration for rb+, the rubidium ion. The aufbau electron configuration method is 1s 2s 2p 3s 3p 4s 3d 4p 5s 4d 5p 6s 4f 5d 6p 7s 5f 6d. Nevertheless, check the complete configuration and other interesting facts about niobium that most people don't know.
What Is Br Full Electron Configuration Written By Rogers Torgartor Tuesday, November 23, 2021 Add Comment Edit.
1s2 2s2 2p6 3s2 3p6 3d10 4s2 4p5. Each element has a unique atomic structure that is influenced by its electronic configuration, which is the distribution of electrons across different orbitals of an atom. (1 point) which atom is larger?
A Bromine Atom Has Two Electrons In Its First Energy Level, Eight Electrons In Its Second, 18 Electrons In Its Third And Seven Electrons In Its Fourth.
The oxidation state of the element changes depending on the bond formation. Full ground state electron configuration for br. Full electron configuration of bromine: